Ever wonder if your friend, the one who always orders the extra spicy tacos, is somehow different from their milder-mannered twin? Well, in the quirky world of atoms, something similar happens. These aren't twins, though. They're called isotopes. And, believe it or not, telling them apart is more fun than you'd think. Forget fancy lab coats and complicated charts. We're talking about everyday detective work, atom-style!
Think about it this way: imagine you have a bunch of identical LEGO bricks. They all look the same, right? But what if some of those bricks had a tiny extra bit stuck to them? They'd still snap into place, but they'd be a little heavier. That's kind of what isotopes are. They're like the same element, but with a slightly different weight. It’s like a celebrity whose lookalike is almost perfect, but you can just tell something's off. Maybe their chin is a millimeter wider, or their laugh is a decibel higher.
So, how do we, the curious onlookers, sniff out these elusive atom-lookalikes? It's all about their number of neutrons. Protons? They're the strict bouncers at the atomic club. They decide who gets in and what element you are. Every atom of, say, carbon, must have six protons. No exceptions! But neutrons? They're the chill party guests. They hang out in the nucleus, minding their own business, and can come and go without changing the element's fundamental identity. But they do add weight. And that's our clue!
"It’s like having a secret handshake for atoms. And we're trying to figure out the handshake."
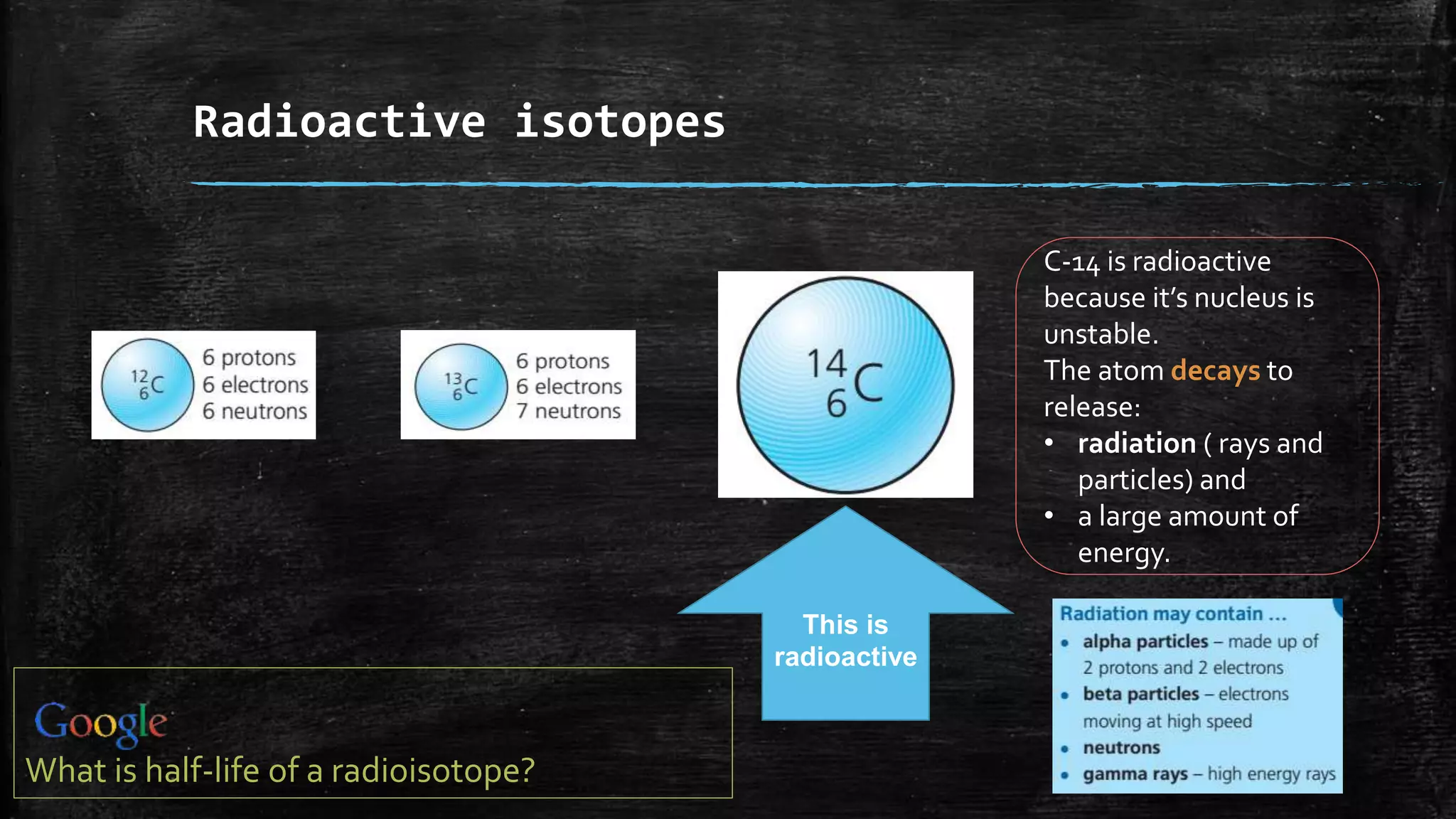
Let's take our buddy carbon again. The most common kind of carbon, the one you find in your everyday life, is called carbon-12. Why 12? Because it has 6 protons and 6 neutrons. Add them up, and voilà! You get 12. Simple math, right? It’s the carbon equivalent of your perfectly average, run-of-the-mill neighbor. Always there, doing its thing, not causing much fuss.
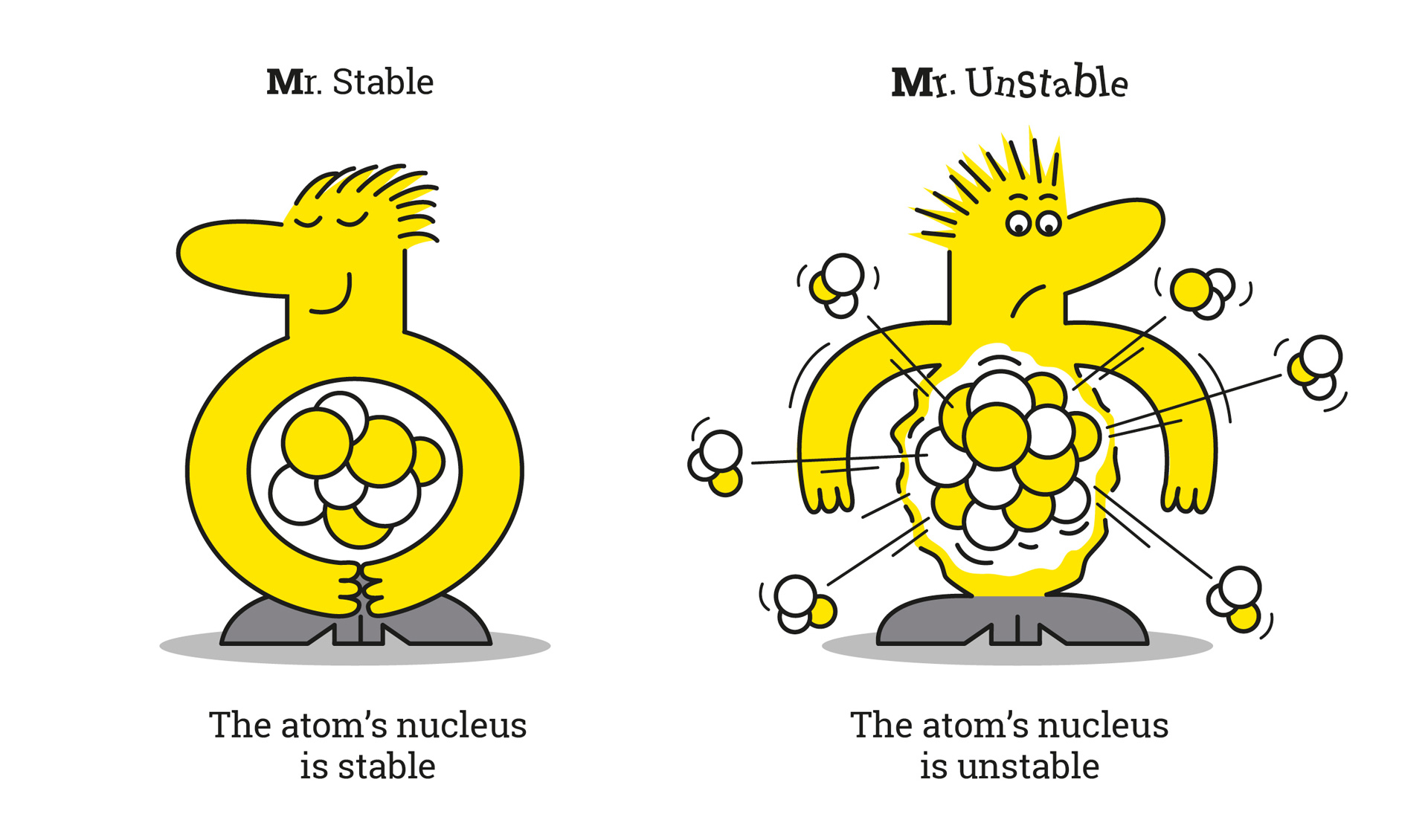
But then, there's carbon-14. This is the cool, slightly eccentric cousin. It also has 6 protons (because it's still carbon, remember the bouncers!), but it has 8 neutrons. So, 6 protons + 8 neutrons = 14. See? It’s heavier. This guy is famous for being used in radiocarbon dating. That's how scientists figure out how old ancient stuff is. It's like carbon-14 has a built-in stopwatch, slowly ticking away its radioactivity. Pretty neat, huh? It’s the carbon that’s been on an adventure, seen things, and has a story to tell.
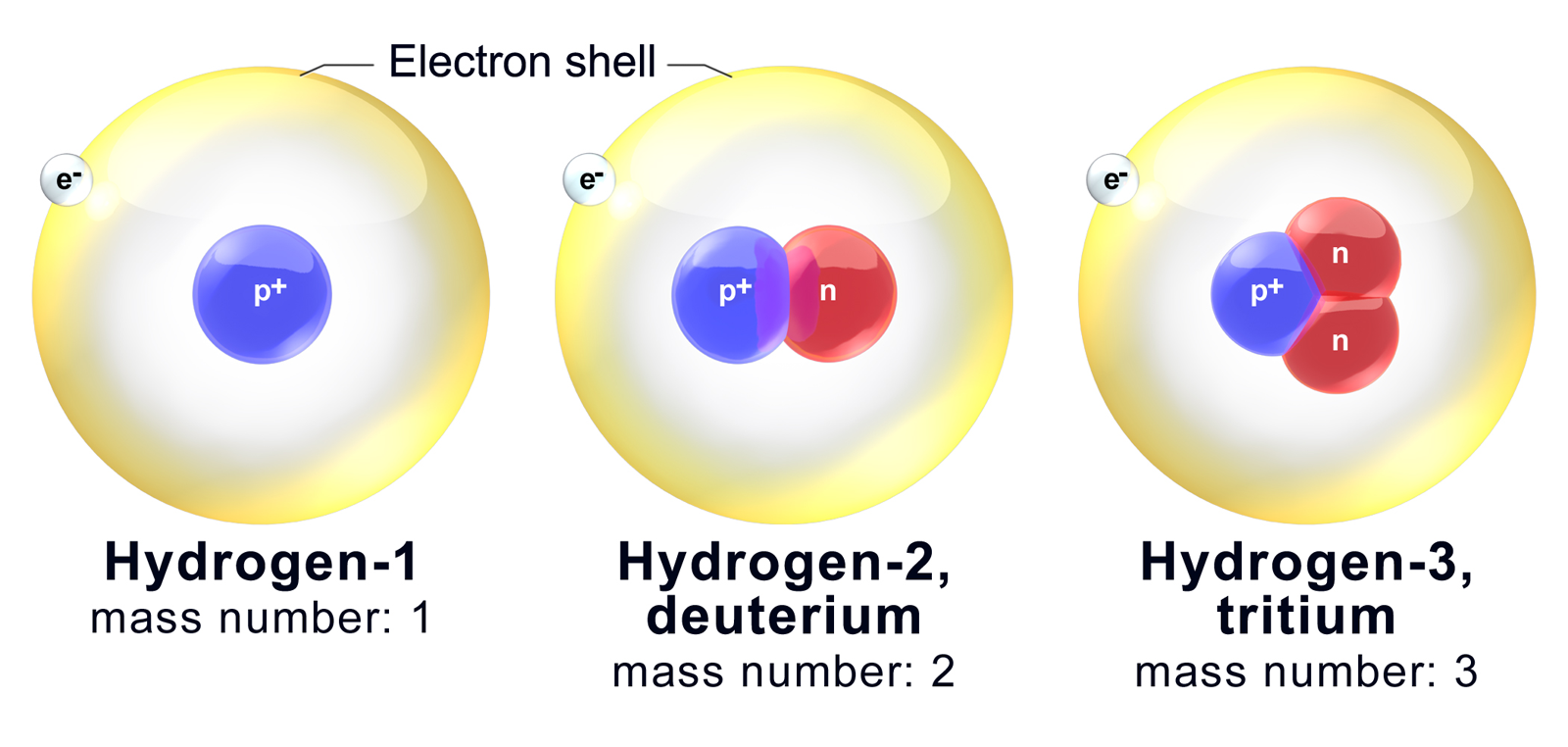
Then there's hydrogen. Oh, hydrogen! The simplest element, the baby of the periodic table. It has just one proton. That’s it. But hydrogen loves to play dress-up with its neutrons. The most common hydrogen, often called protium, has zero neutrons. Just a lonely proton. It's the minimalist hydrogen. Very chic.
But then you meet deuterium. This hydrogen has one neutron. So, 1 proton + 1 neutron = 2. It’s a bit more substantial. Scientists sometimes call it "heavy hydrogen." Imagine your friend who’s always got a backpack on, even when they're just going to the corner store. Deuterium is that friend. It’s just carrying a little extra gear.
And then there's tritium. This hydrogen is the ultimate overachiever. It has one proton and two neutrons! 1 proton + 2 neutrons = 3. It's quite heavy for its size and it's radioactive. Tritium is the daredevil of the hydrogen family. It's the one who jumps off the high dive without a second thought. It's got a lot of energy to release.
So, how do we really tell them apart? We can't just point and say, "Aha! That one's got an extra neutron!" It’s not like they have little name tags. Scientists use clever tools that can detect these subtle differences in mass. Think of it like a very, very precise scale. Or a super-sensitive echo-location system that can tell how many "party guests" are inside the atomic nucleus.
One common method is called mass spectrometry. It sounds intimidating, but picture this: you have a bunch of these isotopes, and you send them whizzing through a magnetic field. The heavier ones (with more neutrons) get bent a little differently than the lighter ones. It's like trying to steer a big, bulky truck versus a zippy little scooter through a curvy road. The truck takes a wider turn. The scooter can zip right around the corner. We can see where they end up, and that tells us their mass, and thus, their identity as an isotope.

Another way is through how they behave in chemical reactions. While the number of neutrons doesn't change what element it is, it can sometimes slightly alter how fast or easily an atom participates in certain reactions. It's like how twins might have slightly different preferences for food, even though they both love pizza. One might prefer extra pineapple, while the other finds that an abomination. Subtle, but noticeable to those who care about such things.
Ultimately, telling isotopes apart is like being a keen observer of the tiny world. It’s about noticing the details that others might miss. It’s not about some grand, earth-shattering difference, but about those little nuances that make each one unique. And isn't that, in a way, a bit like life itself? We're all made of similar building blocks, but the way we're put together, the extra little bits we carry, that's what makes us, well, us. So next time you hear about isotopes, remember they're just atoms with a little extra flair, a slightly different story, and a weightier presence. And that's pretty cool, in our humble, slightly opinionated opinion.