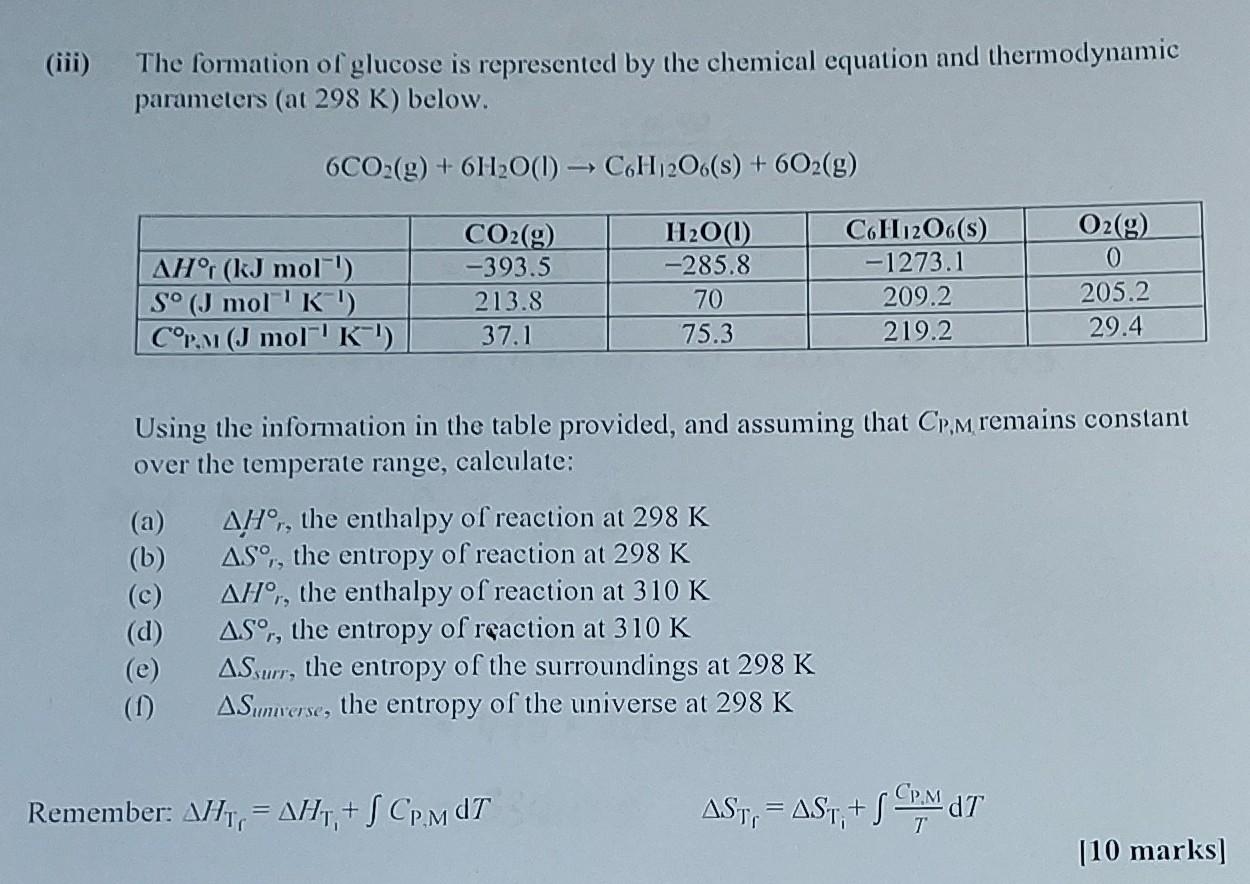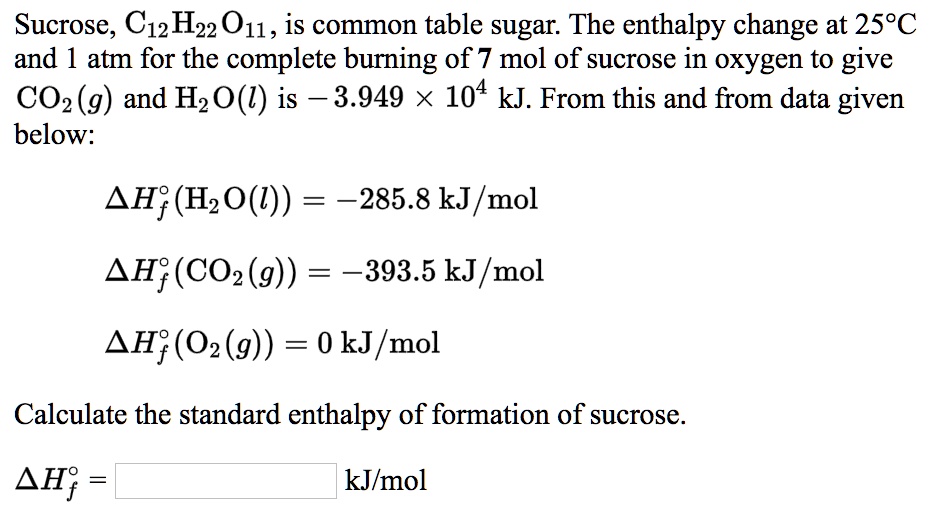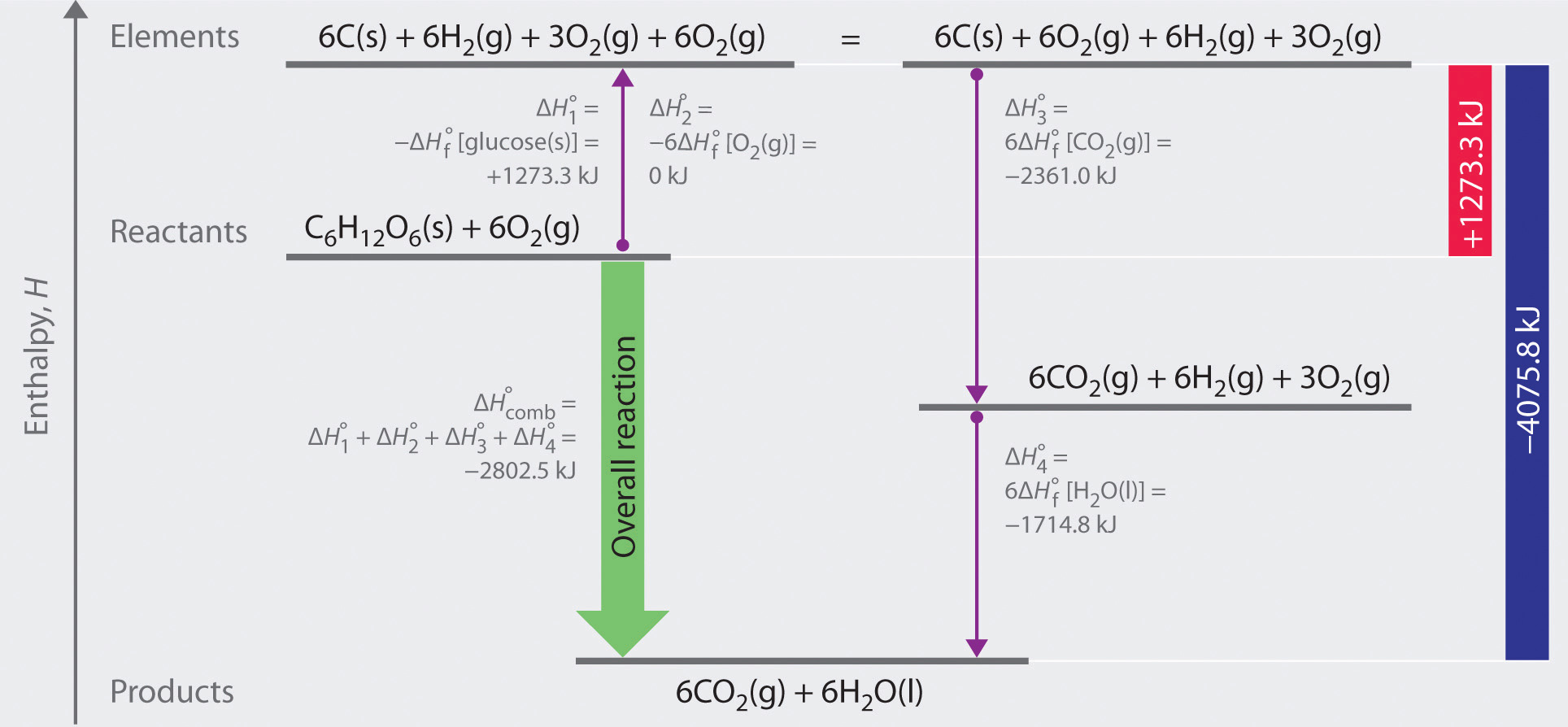
Hey there, science curious folks! Ever wondered about the secret recipe behind that sweet, sweet sugar? Yep, we're talking about glucose, the fuel that powers everything from our brains to our morning coffee. But have you ever thought about how much oomph it takes to put that sugary molecule together?
Well, buckle up, because we're diving into something super cool: the Enthalpy of Formation of Glucose. Sounds a bit fancy, right? But don't let the big words scare you. It's actually a really neat concept that tells us a story about energy!
The Energy Story of Sugar
Think of it like this: building anything, even a tiny sugar molecule, requires effort. You need to gather the building blocks and put them in just the right places. This effort translates into energy. The enthalpy of formation is like the energy price tag for creating one mole of a substance from its basic ingredients.
For glucose, those basic ingredients are its elements in their simplest forms. We're talking about carbon (like in charcoal!), hydrogen (the lightest gas!), and oxygen (the stuff we breathe!). So, the Enthalpy of Formation of Glucose is the energy released or absorbed when one mole of glucose is made directly from pure carbon, hydrogen gas, and oxygen gas.
Why is this so rad?
Because it’s a peek into the fundamental nature of glucose! It tells us how stable glucose is compared to its raw components. Is it an energy-hoarding molecule, or does it happily give up its energy when it’s made?
The value itself is a number, usually in kilojoules per mole (kJ/mol). For glucose, this number is a hefty negative one. What does a negative number mean in this energy game? It means that when glucose is formed from its elements, it actually releases energy!
Imagine building a Lego castle. You put in effort, right? But what if, as you clicked the bricks together, tiny sparks of energy flew off? That's kind of what happens when glucose is born from its elemental parents.

Glucose: A Natural Energy Saver (and Giver!)
This energy release is super important. It tells us that glucose is a relatively stable molecule. It doesn't want to break apart easily. And, more importantly, it means that when our bodies break down glucose, they get to reclaim all that stored energy!
This is the magic behind why we eat! Glucose is our body's favorite quick energy source. When we digest food, we break down complex carbohydrates into simple glucose molecules. Then, our cells can "burn" this glucose to power all our activities.
The negative enthalpy of formation of glucose is the promise of energy waiting to be unleashed. It's like a tightly wound spring, holding a significant amount of potential energy.
A Little Bit of Chemistry Fun
The chemical formula for glucose is C6H12O6. So, we're talking about six carbon atoms, twelve hydrogen atoms, and six oxygen atoms all cozied up together in a specific, sweet arrangement.
To figure out the Enthalpy of Formation of Glucose, scientists don't just magically measure it. They often use something called Hess's Law. This is a clever trick that lets us calculate the enthalpy change for a reaction by using the enthalpy changes of other, known reactions.

It’s like solving a puzzle. If you know the energy cost of getting from point A to point B, and from point B to point C, you can figure out the energy cost of getting directly from point A to point C, even if you can't measure that direct path easily.
The Beauty of Measurement
The measured value for the standard Enthalpy of Formation of Glucose is approximately -1271.3 kJ/mol. That's a lot of energy! To put it in perspective, that's enough energy to do quite a few things.
It's the energy that's stored within the bonds of the glucose molecule. Think of it as the "sweetness" of energy. The more negative the enthalpy of formation, the more energy is released when it's formed, and thus, the more energy is stored within it.
This number isn't just a dry fact for textbooks. It connects the microscopic world of atoms and bonds to the macroscopic world of energy we experience every day. It’s a fundamental constant that underpins life as we know it.

Why is this number so fascinating?
It highlights the efficiency of nature. Plants, through photosynthesis, take simple ingredients (carbon dioxide and water) and, using sunlight, create this energy-rich glucose. The negative enthalpy of formation is a testament to the energy they manage to capture and store.
It’s a beautiful demonstration of how chemical reactions involve energy changes. Some reactions give off energy (exothermic), and some take in energy (endothermic). The formation of glucose is a classic example of an exothermic process, releasing stored energy.
Understanding this value helps us appreciate the energy flow in ecosystems. It's the foundation of food webs, where energy is transferred from producers (plants) to consumers. That stored energy in glucose is the currency of life.
Beyond the Basics
The Enthalpy of Formation of Glucose isn't just about glucose itself. It's a concept that applies to all chemical compounds. Every molecule has its own unique energy story, its own enthalpy of formation.
Studying these values allows chemists to predict how reactions will behave. Will a reaction release heat and be potentially useful for energy production, or will it require a lot of energy to get going?

For glucose, this value is particularly special because glucose is so central to life. It's the direct fuel for our cells, and its formation and breakdown are critical processes.
A Little More Enthusiasm!
So, next time you enjoy something sweet, whether it's a piece of fruit or a spoonful of sugar, remember the incredible journey that molecule took. Remember the energy that was released when it was formed, and the energy it holds, ready to be released for you.
The Enthalpy of Formation of Glucose is more than just a number. It's a window into the energetic heart of life's most fundamental fuel. It's a tiny piece of scientific magic that explains why sugar is so sweet and so important!
Isn't that just batter-y amazing? It’s a sweet deal for our bodies and a fascinating concept for our minds. So go ahead, give a little nod to the energy story of glucose. It’s truly a sweet science!