Alright, so imagine the universe is like a giant, super-organized, and slightly bonkers party. And at this party, there are these two super important guests: electrons and protons. You might have heard of them. They’re basically the Beyoncé and Jay-Z of the atomic world, always in the spotlight, and let me tell you, their relationship is fascinating. So, do they, you know, attract each other? Drumroll please… YES! They totally do!
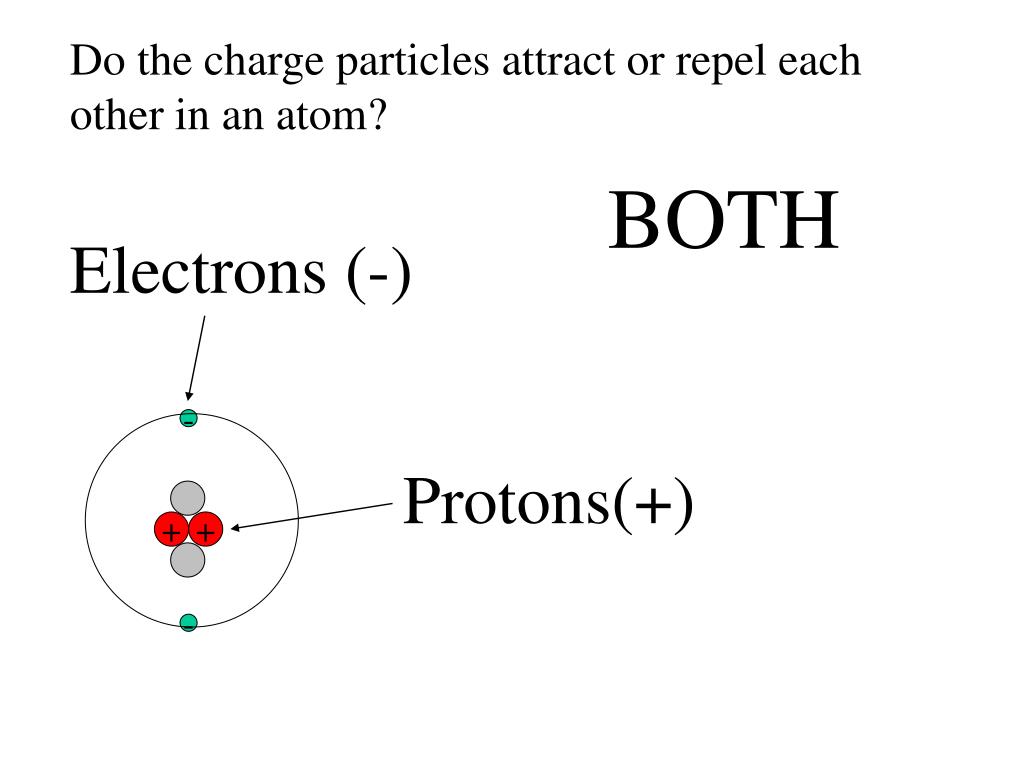
It’s not like they’re holding hands at the punch bowl, mind you. It’s a bit more… scientific. Think of it like this: protons are the super popular kids. They’ve got this positive vibe, this unmistakable “I’m the leader here” aura. You can spot them easily because they’re chilling in the center of the atom, like the VIPs in the penthouse suite. They’re the ones with the positive charge. Like that friend who’s always buying rounds of drinks – generous and, well, positive!
Now, electrons? They’re the total opposite. They’re more like the dancers on the floor, constantly buzzing around, never quite settling down. They’re tiny, speedy little things, and here’s the kicker: they’ve got a negative charge. Think of them as the glitter bomb of the atom, always flitting about. They’re the rebels, the free spirits, the ones who’d rather be anywhere but stuck in one place. And because they’re the opposite of protons, they’re drawn to them like a moth to a very, very bright, positively charged flame.
This whole attraction thing is thanks to something called the electromagnetic force. It’s basically the universe’s way of saying, “Opposites attract, duh!” It’s like that magnetic personality you have, the one that pulls people in. Protons and electrons have that, but on a subatomic level. It’s a fundamental rule of the cosmos, right up there with “don’t feed the bears” and “always wear clean underwear.”
So, why is this so important? Well, this little dance of attraction is what holds atoms together. Without it, everything would just be a chaotic mess of unattached particles. Your coffee mug? Gone. Your cat? Poof! Even the air you’re breathing? Nope. It’s the reason why we have stuff. Seriously, this attraction is the ultimate party planner for the universe.

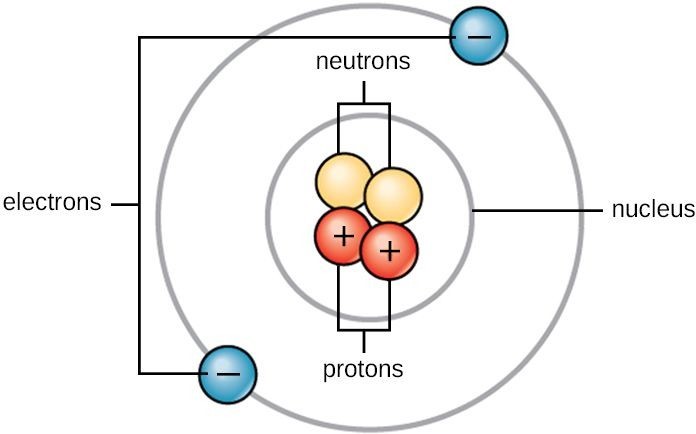
Think of a really simple atom, like hydrogen. It’s got one proton and one electron. That single proton is like the cool host, and the electron is the guest who can't stop circling the dance floor. They’re held together by this invisible tether of attraction. It’s a beautiful, albeit minuscule, friendship. And when you get more protons and electrons involved, it gets even crazier. Like a giant cosmic ballet, but with much tinier dancers and way more invisible forces.
It’s not just attraction, though. Remember how I said electrons are always buzzing around? They don’t just get sucked into the proton’s gravitational pull. Oh no, they’ve got their own thing going on. They’re moving so fast, and they have this thing called momentum. It's like when you're running around a track – you’re being pulled towards the center, but your own speed keeps you from actually crashing into it. Electrons are kind of doing the same thing, perpetually just missing the proton in a graceful, cosmic pirouette.
This delicate balance between attraction and speed is what keeps electrons in their orbits, or more accurately, their electron shells or orbitals. These aren't like the neat, predictable orbits of planets around the sun. Electrons are more like a swarm of bees around a flower, with different probabilities of being found in certain areas. It's a bit fuzzy, a bit unpredictable, and frankly, way more interesting than a perfectly round orbit.
Now, for a little mind-bending fact: protons and electrons aren’t just attracted to each other. Protons also repel other protons. And electrons repel other electrons. It’s like they’re saying, “You’re cool, but not that cool. Let’s keep our distance, buddy.” This repulsion is also part of the electromagnetic force. So, it's not just a love story; it’s a complex social dynamic of attraction and repulsion. Think of it as a cosmic dating app where you’re only allowed to match with someone completely different from you, and you have to aggressively push away anyone too similar.
This dance of forces is what dictates how atoms bond to form molecules. It’s why water is H₂O and not, say, HO₂ or H₃O (although that last one exists under extreme conditions, but let’s not go there). The way electrons are shared or transferred between atoms is all about satisfying these attractions and repulsions, trying to reach a state of maximum chill, or lowest energy, if you want to get fancy.
And here's another wild thought: if you could somehow get rid of all the empty space inside atoms, you and I would be small enough to fit on the head of a pin. Seriously! Most of an atom is just empty space where these little electron dancers are whizzing around. The protons and electrons themselves are incredibly tiny. So, the fact that we’re not collapsing into a singularity is a testament to the incredible power of these fundamental forces, especially the electromagnetic one that keeps our atoms from saying, “Peace out!”
So, next time you’re looking at a table, or a chair, or even your own hand, remember the incredible, ongoing attraction between electrons and protons. It’s a silent, invisible force that’s holding everything together. It’s the ultimate cosmic glue, the reason for existence, and frankly, one of the most entertaining partnerships in the entire universe. They might be tiny, and they might be part of something you can’t even see, but their attraction is what makes everything possible. Pretty neat, huh?