Ever feel like some people just… spark? Like, they’ve got this extra zest, this little bit of zing that makes them stand out? Well, believe it or not, atoms can be a bit like that too. And when we’re talking about atoms with halogen characteristics, we’re talking about the chemical equivalent of those friends who are always up for an adventure, or maybe a little bit… clingy. You know the type. The ones who really want to join the party, and they'll do whatever it takes to make sure they're included.
Think of it like this: imagine you’ve got a bunch of atoms chilling out, minding their own business. Most of them are pretty content with their current situation. They’re like folks who are perfectly happy with their established routine, their comfy armchair, and a good book. But then you’ve got these special atoms. They’re the ones who are always peeking over the fence, eyeing up what their neighbors have. Specifically, they’re looking for one more electron. Just one. It’s like they’ve got a single empty slot in their mental address book, and they’re desperately trying to fill it.
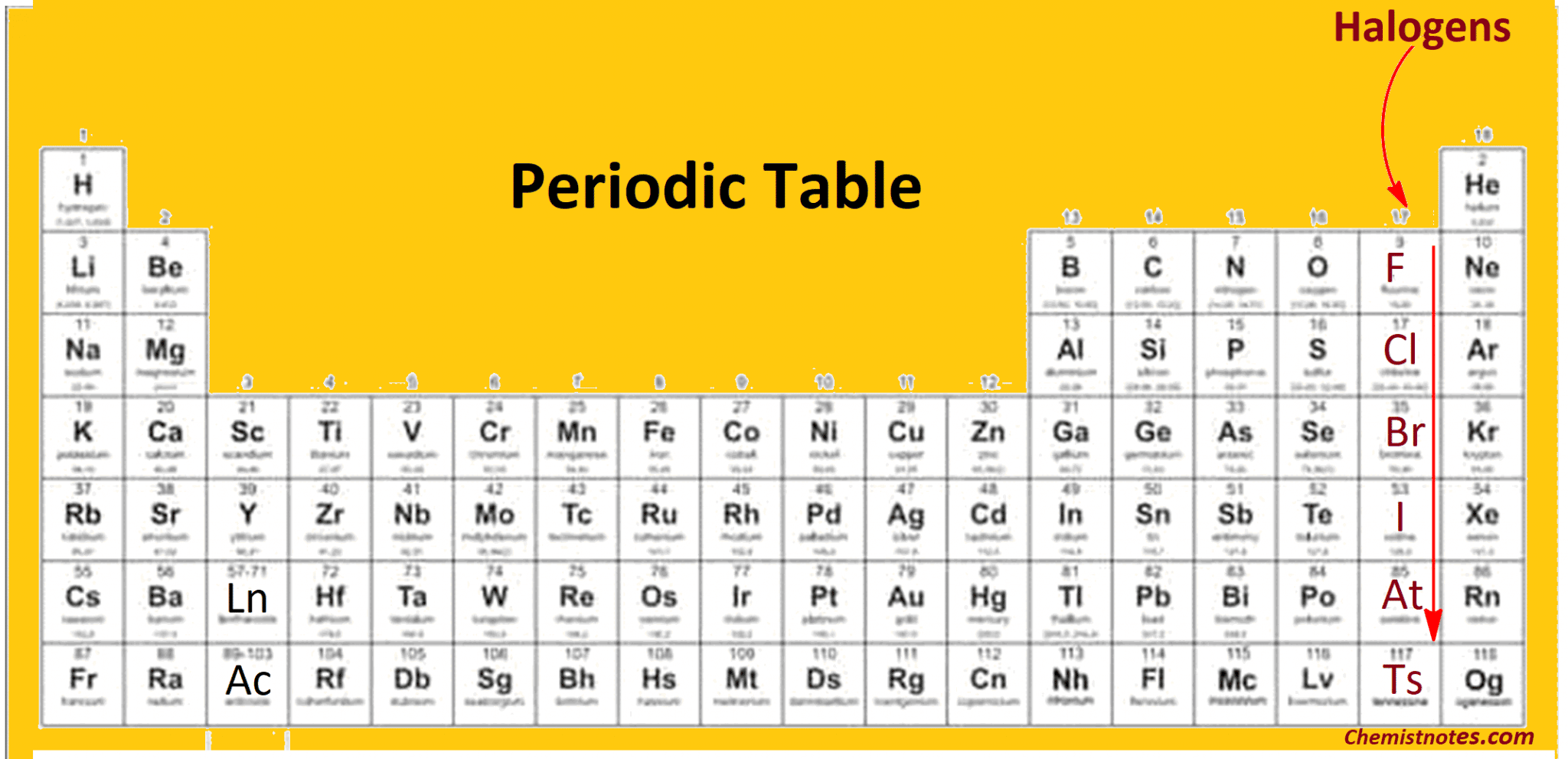
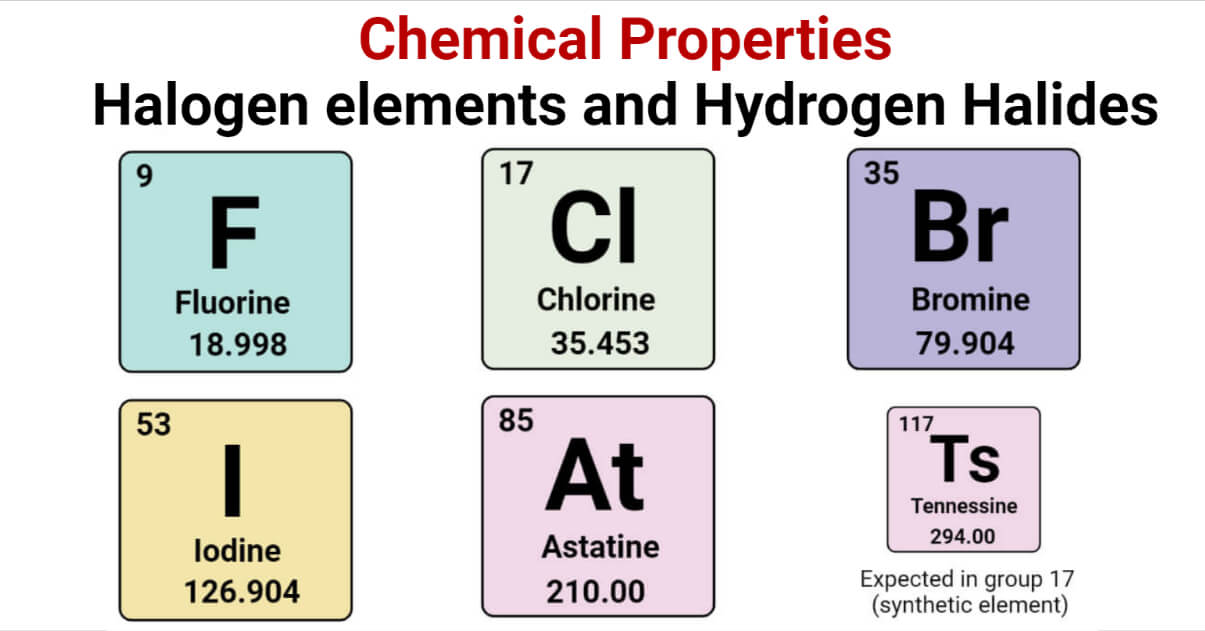
These are our halogens. They’re the elements in Group 17 of the periodic table, and if you’ve ever heard of fluorine, chlorine, bromine, iodine, or astatine, you’ve met some of these guys. They’re the ultimate social butterflies of the atomic world, always looking to bond with someone. And when I say bond, I mean really bond. They're not just looking for a casual acquaintance; they're looking for a committed, long-term relationship. Like, "We're in this together, come what may" kind of commitment.
So, why are they so eager to grab that extra electron? It all comes down to something called stability. Atoms, bless their tiny little hearts, are always striving for a full outer electron shell. It’s like a perfectly organized sock drawer – everything has its place, and nothing is left out. For most atoms, having eight electrons in their outermost shell (we call this the octet rule) is the golden ticket to happiness and stability. It’s the atomic equivalent of achieving inner peace. And our halogens? They’re just one electron shy of that ultimate chill-out state. They're like the person who’s one step away from finishing a marathon, or one ingredient away from making that perfect recipe. So close, yet so… electron-less.
This desperate need for an electron makes them incredibly reactive. They’re not shy about seeking out partners. Imagine a really popular kid at school who’s looking for a dance partner at the prom. They’re going to be actively searching, maybe even a little bit intense in their pursuit. Halogens are like that, but on a subatomic level. They’ll happily snatch an electron from another atom if it means achieving their own stable state. It’s a bit like a really enthusiastic host at a party who insists you have another slice of cake, even if you’re already full. They just want to make sure everyone’s… satisfied.
Let’s take fluorine, the undisputed champion of electron-snatching. Fluorine is so incredibly electronegative – meaning it has a ridiculously strong pull on electrons – that it’s practically a black hole for them. It’s like the ultimate bargain hunter who sees something on sale and has to have it, no matter what. It’ll take an electron from pretty much anything it can get its hands on. Seriously, it's the atom equivalent of someone who buys the last packet of their favorite biscuits at the shop. They’ve secured their prize!
Then you’ve got chlorine. Chlorine is probably the most famous halogen for most people, thanks to its role in everyday life. You know, the stuff they put in swimming pools to keep things clean and sparkling? That’s chlorine at work. It’s a disinfectant, a bleach, and it’s pretty good at its job. It’s like a diligent cleaner who comes in and makes everything sparkle. And it does this by being reactive, by interacting with… well, stuff. It grabs onto things, often in a way that makes them less hospitable to unwanted germs. It’s a bit like that friend who’s always tidying up your messy room for you, whether you like it or not.
Bromine is another one. Bromine is a liquid at room temperature, which is already a bit unusual. It’s reddish-brown and has a rather… pungent smell. Think of it as the slightly mysterious, possibly moody cousin in the halogen family. It’s reactive, but perhaps a tad less aggressive than fluorine. It’s more like someone who will still offer you a piece of cake, but maybe after a gentle hint and a knowing glance.
Iodine, on the other hand, is the gentler giant. It’s a solid at room temperature, and it’s actually essential for our health! We need it for our thyroid function. That little brown stuff they put on a scrape? That’s often iodine. It's the halogen that’s trying to help out. It’s like the wise elder who’s still active and contributing, but in a more measured way. It’s still looking for that electron, but it's not going to go around causing a ruckus to get it.

What’s really fascinating is how these halogen characteristics lead to the formation of salts. When a halogen atom finally snags that extra electron, it becomes a negatively charged ion (an anion). Often, it’ll grab that electron from a metal atom, which then becomes a positively charged ion (a cation). These oppositely charged ions are then attracted to each other, like magnets. And boom – you’ve got a salt. Table salt, for instance, is sodium chloride. Sodium is a metal, and chlorine is our reactive halogen friend. Sodium is happy to give up an electron, and chlorine is thrilled to take it. They get together, form an ionic bond, and create something we use every single day. It’s the ultimate atomic matchmaking!
It’s like when you’re at a party and everyone’s mingling, and then two people just click. They hit it off instantly, and suddenly they’re inseparable. That’s what’s happening with sodium and chlorine. The metal atom is like the generous donor, and the halogen is the eager recipient. And the result? A stable, delicious, and utterly essential compound.
But halogens aren't just about making salt. Their reactivity is crucial for a whole bunch of industrial processes. They're used in everything from plastics to pharmaceuticals. Think about Teflon, that super slippery stuff on your non-stick pans. That’s a fluorinated polymer. Fluorine, our electron-hoarding superstar, is a key ingredient. It’s like the secret sauce that makes things… work. Without these reactive little guys, a lot of the modern conveniences we take for granted wouldn’t exist.
Imagine trying to make a cake without flour, or a sandwich without bread. That’s kind of what our modern world would be like without the unique properties of halogens. They’re the essential building blocks, the indispensable ingredients. They might seem a bit pushy in their pursuit of electrons, but their enthusiasm is what drives so many chemical reactions.
It’s important to remember that while halogens are essential, they can also be quite hazardous in their pure form. Chlorine gas, for example, is toxic. Bromine fumes are irritating. This is because they are so reactive. They can readily interact with the molecules in our bodies in ways that are not beneficial. It’s like that friend who’s too enthusiastic – their energy can be a bit overwhelming if not managed properly. They need to be handled with care and respect, just like any powerful chemical.
This is why we don’t just go around playing with pure chlorine or bromine. We use them in controlled environments and in compounds where their reactivity is harnessed for good. It’s like taking that high-energy friend and channeling their enthusiasm into a productive project, rather than just letting them bounce off the walls. The key is to understand their nature and use it wisely.
The characteristic electron affinity of halogens is what makes them so unique. They have this inherent drive to gain an electron, which is a powerful force in chemistry. It's what makes them such excellent oxidizing agents – they readily accept electrons from other substances, causing those substances to be oxidized. It’s a bit like a battery, where one component gives up electrons and another accepts them, creating a flow of energy.
So, the next time you’re sprinkling salt on your fries, or admiring the pristine blue of a swimming pool, or even just using a non-stick pan, take a moment to appreciate the humble, yet incredibly powerful, halogen. They might be a bit demanding in their quest for electrons, but their characteristic zest is what makes our world a more stable, more functional, and dare I say, tastier place. They are the little atoms with big personalities, always ready to make a connection, and in doing so, they shape the very world around us.
They’re the unsung heroes of the periodic table, the ones who are always willing to go the extra mile (or, in this case, grab the extra electron) to achieve balance and create something new. And honestly, who can’t relate to that? We all want to feel complete, to have our electrons in order, and to form meaningful bonds. The halogens are just a little more explicit about it.

/periodic-table-935378844-5c52765bc9e77c0001380acb.jpg)