Hey there, you! Ever wondered what goes on in the teeny-tiny world of atoms? You know, those building blocks that make up everything? Well, today we're gonna have a little chat about one of the coolest kids on the atomic block: an Alkali Metal. Think of them as the rockstars of the periodic table, always ready to party and make some serious chemical noise!
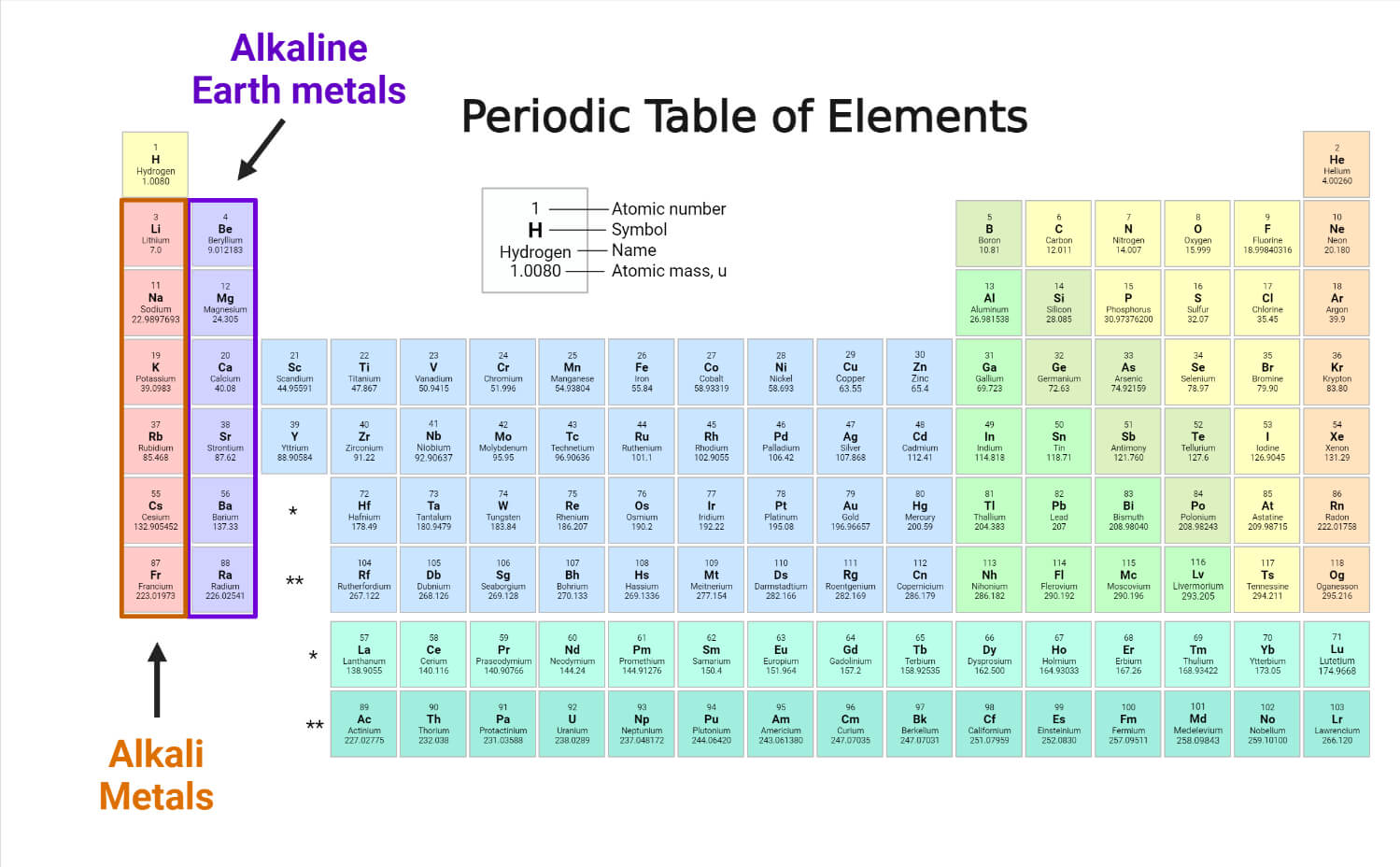
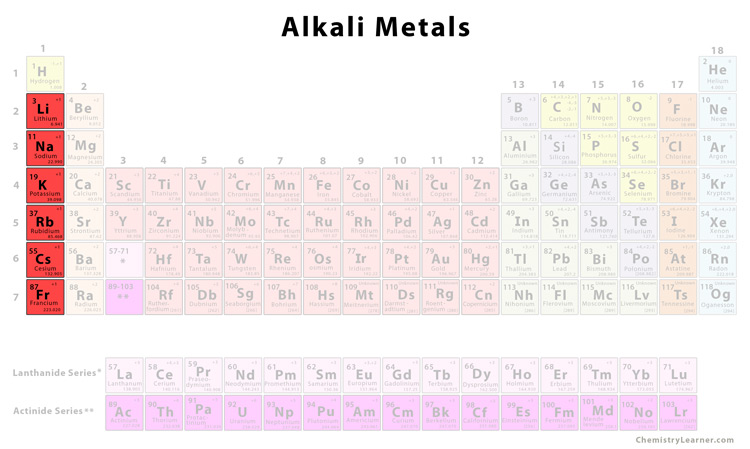
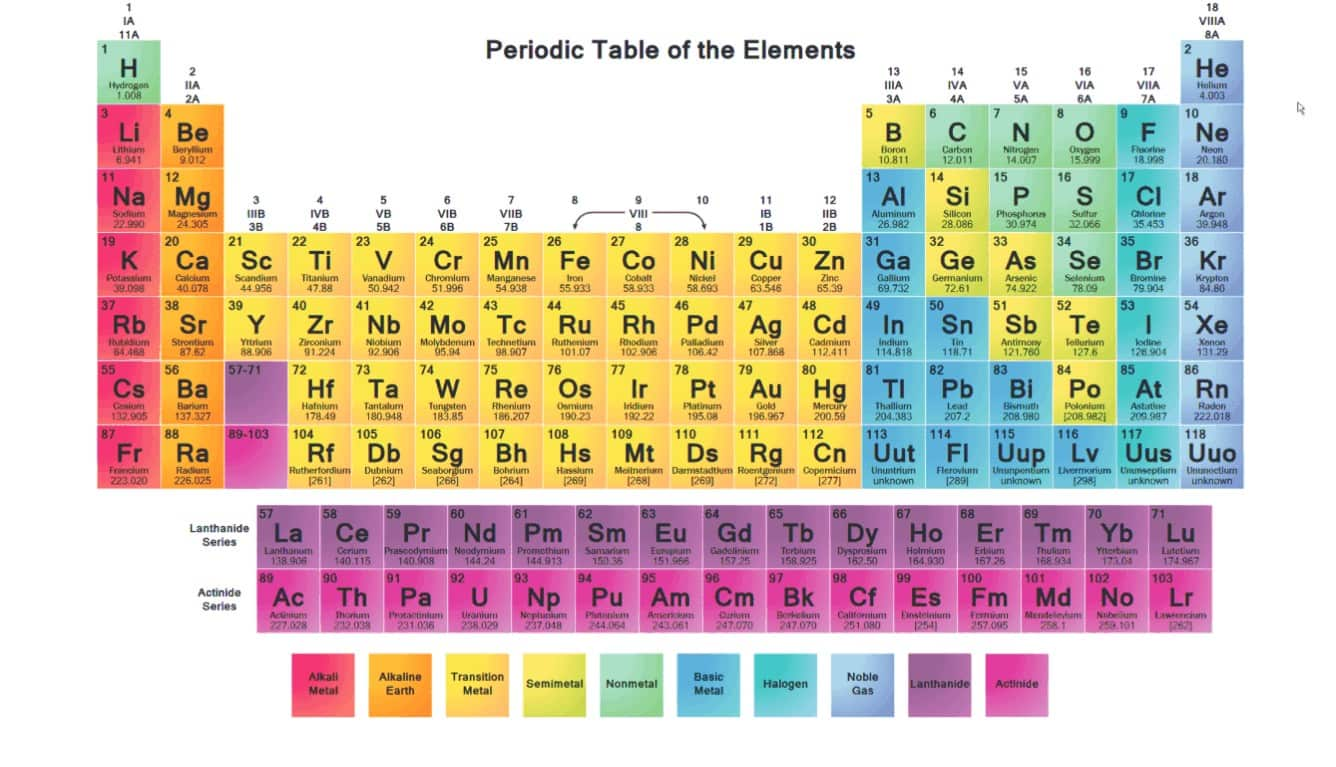
So, what exactly IS an Alkali Metal? Imagine the periodic table, that giant chart of all the elements. Our Alkali Metals are chilling in the very first column on the left. That's column number 1, folks! They're the VIP section, the front row seats. Pretty neat, right?
These guys are special. Like, really special. They've got this one extra electron hanging out in their outermost shell. It's like having a spare key to a super-exclusive club. This little electron is just itching to get out there and do its thing. It's a bit of a rebel, if you ask me!
Because of this super-reactive electron, Alkali Metals are famously… well, reactive. Like, super reactive. We’re talking about elements that are practically begging to hook up with other elements. They’re the ultimate social butterflies of the atomic world. You can't just leave them lying around, or they might get into trouble. Oops!
Think of it this way: that extra electron is like that one friend who's always up for an adventure. They’re not content to just sit around; they need to explore, to connect, to make something happen. And boy, do they make things happen!
One of the most famous Alkali Metals is Sodium. Yep, that's the stuff in your table salt! But don't get any ideas about popping pure sodium into your fries. Trust me, it's a *terrible idea. Pure sodium? It reacts with water so violently, it's like a mini-firework show. *Poof! It’s exciting, but definitely not for culinary purposes. So, maybe stick to the salt shaker, eh?

Then there's Lithium. You know, the stuff in your phone battery? That's our little lithium buddy at work! It’s a bit lighter than sodium, and it’s also super eager to share that electron. It’s a go-getter! Imagine a tiny, energetic little dude zipping around, powered by lithium. That’s your phone!
And let's not forget Potassium. This one is crucial for keeping your body humming along nicely. It’s involved in all sorts of important jobs, like muscle contractions and nerve signals. So, thank potassium next time you wave hello or do a little dance. It’s working hard behind the scenes!
Alkali Metals are also known for being quite soft. Like, really soft. You could probably slice them with a butter knife. Imagine cutting through metal with a butter knife! It's kind of a mind-blowing thought, isn't it? They’re not your tough, rugged metals; they're more like the approachable, laid-back members of the metal family.
This softness, combined with their extreme reactivity, means you usually don't find them hanging out in their pure form in nature. They're too busy reacting with everything! They're like those popular kids who are always in a group; you rarely see them alone. They're typically found bonded with other elements, forming compounds.

For example, sodium and chlorine – our table salt buddies – are a perfect match made in chemical heaven. Sodium, with its eagerness to lose an electron, and chlorine, with its eagerness to grab one, just *click. It’s a beautiful chemical romance.
Let's dive a bit deeper into why they're so darn reactive. Remember that one electron in the outer shell? It's like a single sock without a mate. It just feels incomplete. It *really wants to find a friend, another atom to bond with. And when it finds one, BAM! A chemical reaction happens. They're like the ultimate matchmakers of the atomic world.
This strong desire to get rid of that electron is what makes them such good reducing agents. Fancy term, I know! Basically, it means they're really good at giving away electrons to other atoms. They're the generous givers of the periodic table. Always sharing, always contributing. What good pals!
Because they're so reactive with water, Alkali Metals are usually stored under oil. Yep, oil! It’s their little way of staying safe and preventing an accidental, fiery reunion with H₂O. Imagine your phone battery floating in a pool of oil to keep it from exploding. A bit dramatic, but it works!
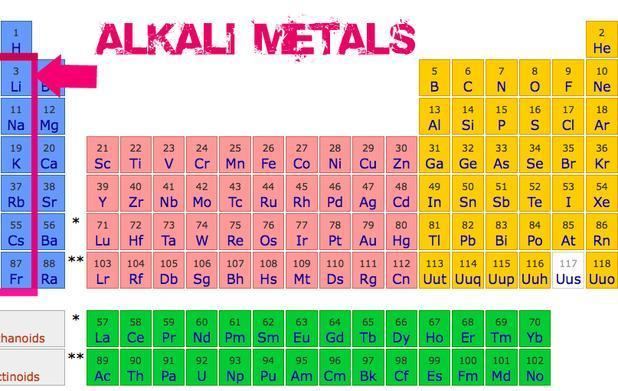
The reactivity increases as you go down the column. So, lithium is pretty reactive, sodium is more reactive, potassium is even more reactive, and it just keeps going. By the time you get to the bottom, like Francium, you've got an element that's so unstable and reactive, it barely hangs around long enough for us to study it properly. It's like the shyest kid in class who's always hiding behind the teacher. Poof! Gone before you know it.
Don't let their reactivity fool you, though. Alkali Metals play a huge role in our lives. Besides sodium in salt and lithium in batteries, potassium is in bananas and potatoes (your heart will thank you for those!). And even though they're not usually found in their pure form, their compounds are everywhere.
Think about cleaning products. Many of them contain compounds of alkali metals. They help to break down grease and grime, making our lives a little bit cleaner. So, next time you’re scrubbing away, give a nod to those hardworking alkali metal compounds. They’re the unsung heroes of cleanliness!
They also play a vital role in industry. For instance, sodium is used in the production of certain metals and in manufacturing paper. Lithium is not just for batteries; it's also used in making glass and ceramics more durable.

And let's not forget the vibrant colors they can create! When heated, some alkali metals produce beautiful flames of color. Sodium gives off a bright yellow flame, potassium a lilac or pale violet one, and lithium a brilliant red. It’s like a mini fireworks display brought to you by chemistry! Imagine a whole party lit up by these colorful atomic sparks. That would be something!
So, here we have our Alkali Metals: a bunch of soft, shiny, super-reactive elements that are incredibly important. They might be a handful to handle in their pure form, but their impact on our world is undeniable. They're the energetic, enthusiastic, and essential building blocks that help make so many things possible.
They remind us that sometimes, the things that seem a little wild or untamed are actually the most powerful and vital. They're not afraid to be themselves, to reach out, and to connect. And in their own explosive, energetic way, they contribute to the beauty and functionality of our universe.
So, the next time you think about atoms, give a little cheer for the Alkali Metals. They’re the friendly, flamboyant stars of the chemical show, always ready to react and make the world a more interesting, and functional, place. And isn’t that a wonderfully uplifting thought? Keep shining, little atoms!